Stability and Sensitivity Analysis of Dengue-Malaria Co-Infection Model in Endemic Stage
Keywords:
Coinfection, Dengue, Malaria, Sensitivity AnalysisAbstract
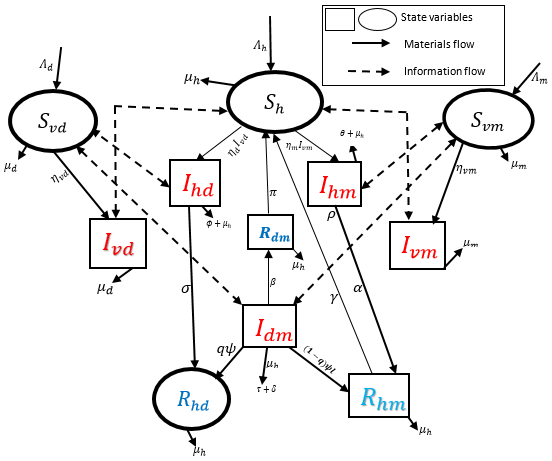
In this study, a deterministic co-infection model of dengue virus and malaria fever is proposed. The disease free equilibrium point (DFEP) and the Basic Reproduction Number is derived using the next generation matrix method. Local and global stability of DFEP is analyzed. The result show that the DFEP is locally stable if R0dm < 1 but may not be asymptotically stable. The value of R0dm calculated is 19.70 greater than unity; this implies that dengue virus and malaria fever are endemic in the region. To identify the dominant parameter for the spread and control of the diseases and their co-infection, sensitivity analysis is investigated. From the numerical simulation, increase in the rate of recovery for co-infected individual contributes greatly in reducing dengue and malaria infections in the region. Decreasing either dengue or malaria contact rate also play a significant role in controlling the co-infection of dengue and malaria in the population. Therefore, the center for disease control and policy makers are expected to set out preventive measures in reducing the spread of both diseases and increase the approach of recovery for the co-infected individuals.
Published
How to Cite
Issue
Section
Copyright (c) 2021 Journal of the Nigerian Society of Physical Sciences

This work is licensed under a Creative Commons Attribution 4.0 International License.





