The corrosion characteristics of SS316L stainless steel in a typical acid cleaning solution and its inhibition by 1-benzylimidazole: Weight loss, electrochemical and SEM characterizations
Keywords:
Corrosion inhibitor, Stainless steel, Acid cleaning, Imidazole, PolarizationAbstract
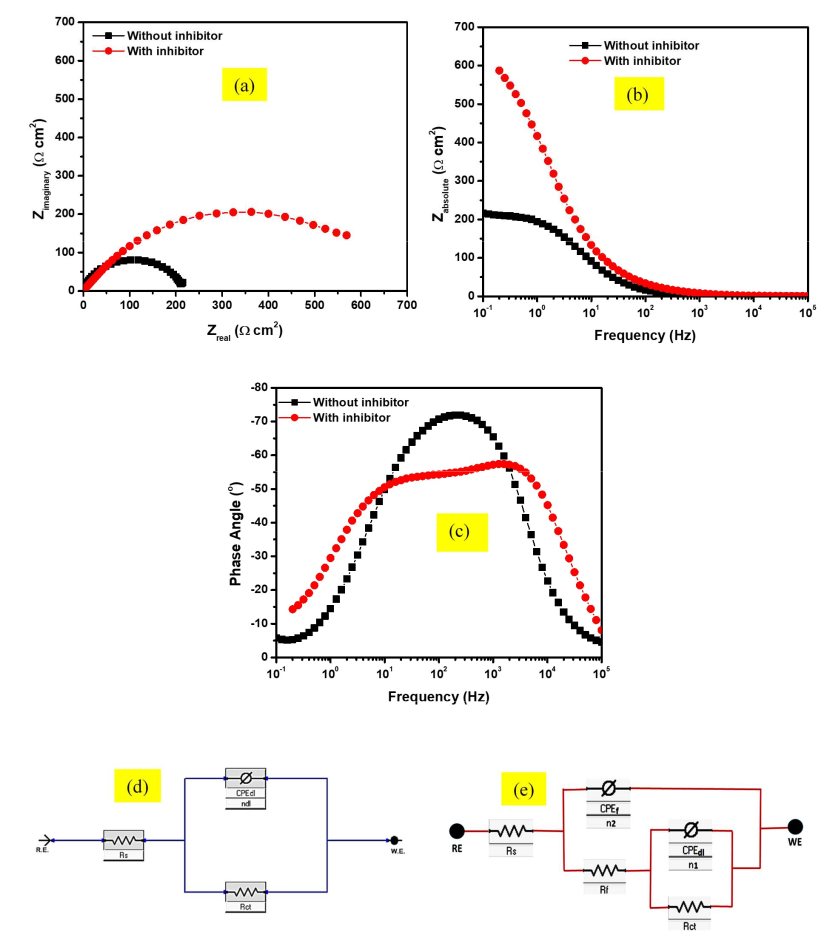
Acid cleaning, an inevitable industrial practice used to descale chemical reactors, usually causes serious corrosion attack on underlying alloy substrates. Ameliorating this phenomenon requires the addition of effective corrosion inhibitors into the acid solution. Current global regulations encourage environmentally–benign molecules as corrosion inhibitors. Consequently, 1-benzylimidazole has been investigated for its inhibitive characteristics against the corrosion of SS316L stainless steel in a typical acid cleaning solution containing 2 % HCl + 3.5 % NaCl. Weight loss measurements confirm that the corrosion inhibition property of 1-benzylimidazole increases with concentration but depreciates with increased temperature. Electrochemical impedance spectroscopy (EIS) and potentiodynamic polarization (PDP) measurements confirm that 1-benzylimidazole adsorb on the stainless steel surface to isolate its surface from the acid solution. 1-benzylimidazole is a mixed-type inhibitor with greater anodic influence, and its adsorption enhances the formation and protectiveness of a passive film. Weight loss and the electrochemical measurements agree to an average inhibition efficiency > 70 % at 1000 ppm. The inhibitor adsorbs via physisorption and obeys the Temkin isotherm model. SEM surface characterization confirm the ability of 1-benzylimidazole to protect the surface microstructure of the stainless steel during the corrosion.
Published
How to Cite
Issue
Section
Copyright (c) 2022 Journal of the Nigerian Society of Physical Sciences

This work is licensed under a Creative Commons Attribution 4.0 International License.





